SMA projects ongoing
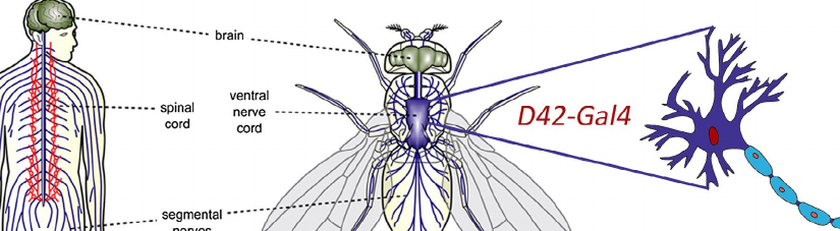
The GT group has used an in vivo SMN2 minigene reporter system in Drosophila motor neurons that serves as a cost-effective, feasible, and stringent primary screening model for identifying chemicals capable of crossing the conserved Drosophila blood-brain barrier and modulating exon 7 inclusion. The model was used for the screening of 1100 drugs from the Prestwick Chemical Library, resulting in 2.45% hit rate. The most promising candidate drugs were validated in patient-derived fibroblasts where they proved to increase SMN protein levels and in mouse models (see results above)
Among them, repurpose moxifloxacin and a second undisclosed drug have been confirmed as modulators of SMN2 splicing by promoting exon 7 inclusion. The promising results after Drosophila screening and validation in relevant mammalian models have resulted in different exciting development possibilities for the project focusing in the developing new treatments for SMA patients
- CAIXAIMPULSE VALIDATE - link here. In recent years, three drugs have been approved to treat SMA, but all have medical limitations for use in the less severe forms of the disease in adults and are extremely expensive. Furthermore, the consequences of its prolonged use are unknown. The GT team has identified two drugs that have been shown to have an excellent safety profile in chronic use. Furthermore, preclinical validation of type II SMA in mice has shown that it favors an outstanding recovery of motor functions and an increase in the levels of the SMN protein in the spinal cord. The objective of the CaixaImpulse Validate is to explore the repositioning of at least one of these drugs and to validate their use in patients with SMA.
- INNOSALUD - link here. R-Drugs is an initiative to enhance the reuse of drugs for rare diseases, starting with two candidate drugs for spinal muscular atrophy (SMA), both FDA-approved drugs with the potential to become an affordable therapy for patients with limited access to expensive existing treatments. SMA is a neurodegenerative disease. The high cost of the treatments (more than 200,000 euros per patient / year) represents a great economic stress for the national health system and private insurers.
- AGENCIA VALENCIANA DE LA INNOVACIÓ (AVI) - The GT group has been awarded with 0,5 M Euros (project INNVA1/2021/44) to perform a clinical trial to evaluate the therapeutic potential of a repurposed drug in SMA patients.